OUR LEADERSHIP TEAM
METAGENICS GLOBAL GROUP
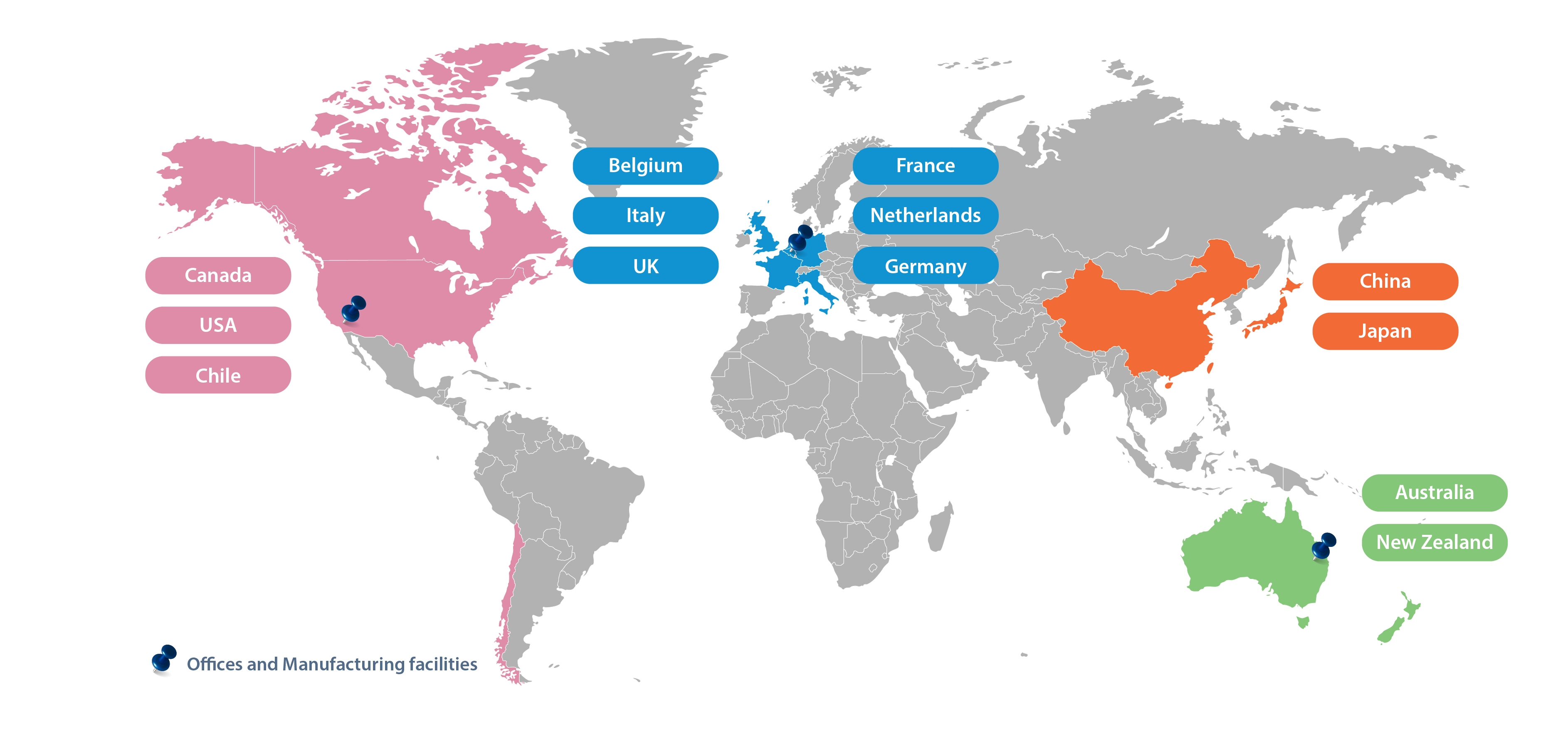
With 1200+ staff globally and presence in every major market in the world, our global footprint continues to expand.
OUR HISTORY
The Metagenics ANZ Story
The Metagenics story began in 1983 when concerned industry advocate, Jeff Katke, tested various major US nutritional suppliers' products and found that they were using low-grade ingredients and reactive excipients (in order to increase their profit margins without any concern for patients' health). Jeff decided to start his own ethical health science company, Metagenics Inc, which would produce only high quality nutritional products to be administered by properly trained Healthcare Professionals.
Under the banner of Metagenics, Leon Brosnan, a retired Pharmacist, and his son Michael, formed Health World Pty Ltd in Australia in 1985. Both had a dream of building a company that would provide high quality nutritional supplements to Healthcare Professionals and consumers.
Renamed Metagenics in 2017, Metagenics has grown to become a market leader and one of the most trusted suppliers of Natural Medicines in Australia and New Zealand. Much of Metagenics's success is due to its strict adherence to its mission of helping people live happier, healthier lives.
Since the company's formation over 35 years ago, Metagenics's quality processes have continued to set new standards in safety and efficacy, and will continue to do so into the future. Metagenics - Australia's and New Zealand's Leading Natural Health Science Company, helping people live happier, healthier lives.

QUALITY AND SAFETY
The Metagenics Quality Process
We know that the clinical benefit delivered by any natural medicine begins with the quality and strength of its raw materials.
At Metagenics, we formulate evidence-based supplements that we test ourselves, from product development through the manufacturing process, and continue to monitor even after the finished product has been released. This ensures the efficacy of our products matches our practitioner’s expectations. Metagenics’ unparalleled commitment to quality means we rigorously vet our raw material suppliers and select only high quality ingredients supported by:
• Scientific research which proves clinical efficacy;
• Thorough assessment for adulterants or substitutions;
• Toxicological analysis including checks for heavy metals, pesticides and microbial contaminants
These meticulous testing procedures ensure that we’re delivering consistently high quality, pure, clean and safe products.
Meet Michael, Director - Head of Regulatory Affairs and Technical
Meet Taryn, Metagenics Product Development Manager.



Selection of the Highest Quality Ingredients Based on Levels of
Scientific and Clinical Research
Safety and Toxicology
Active Constituents
Heavy Metals
Pesticides
Ingredient Quality Control Checks
- Microbial Contamination
- Qualitative Identity
- High Performance Thin Layer Chromatography
- Atomic Absorption
- Gas Chromatography
- High Performance Liquid Chromatography
Manufacturing Standards that Exceed Industry GMP
Low Moisture
Low Heat
Natural Origin, Low Allergy Excipients
Low Excipient Formulas: Metagenics carefully researches any excipients used in our formulas and use safe, low reactive ingredients
Finished Product Analysis
Microbial Contamination
Quantitative Analysis of Actives
Disintegration Time
Weight and Size Uniformity
Moisture Content (stability)
Safety Standards
Metagenics Australia is committed to ensuring the health and safety of not only employees, but everyone who comes in contact with the organisation, by integrating safe working practices across the business.
We believe there is no higher business priority than Occupational Health and Safety and aim to achieve zero harm at all times. Our continuous improvement philosophy, on-going training, as well as implementation and review of targets, plans and procedures, means we are constantly refining workplace safety and health standards.
With a dedicated Workplace Health and Safety function to ensure we comply with all relevant Occupational/Workplace Health and Safety legislation as well as representatives across the business, we have the resources and support network for a safe, healthy and hazard free environment.








